Striving for excellence in healthcare for everyone, everywhere.
For more than 20 years, we've been providing sophisticated, cost-effective solutions that help people lead healthy lives.
Designing the future of healthcare—today
We needed a system or platform that had flexibility and the agility to meet ever-growing and changing demands for our users, and with this platform we now have that.


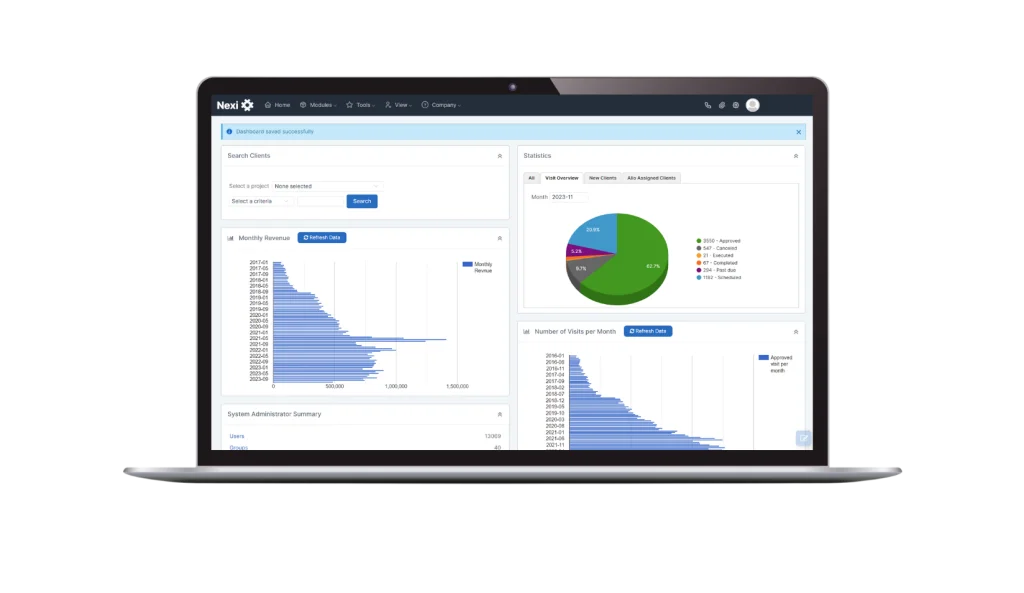
Healthcare software solutions
Helping you increase efficiencies, protect your critical systems and enable new pathways to better care.
Unleash the potential of your healthcare business
Nexi is the innovative healthcare delivery platform that uses automation and analytics to help you deliver more timely, more integrated and more profitable home care and home health services.
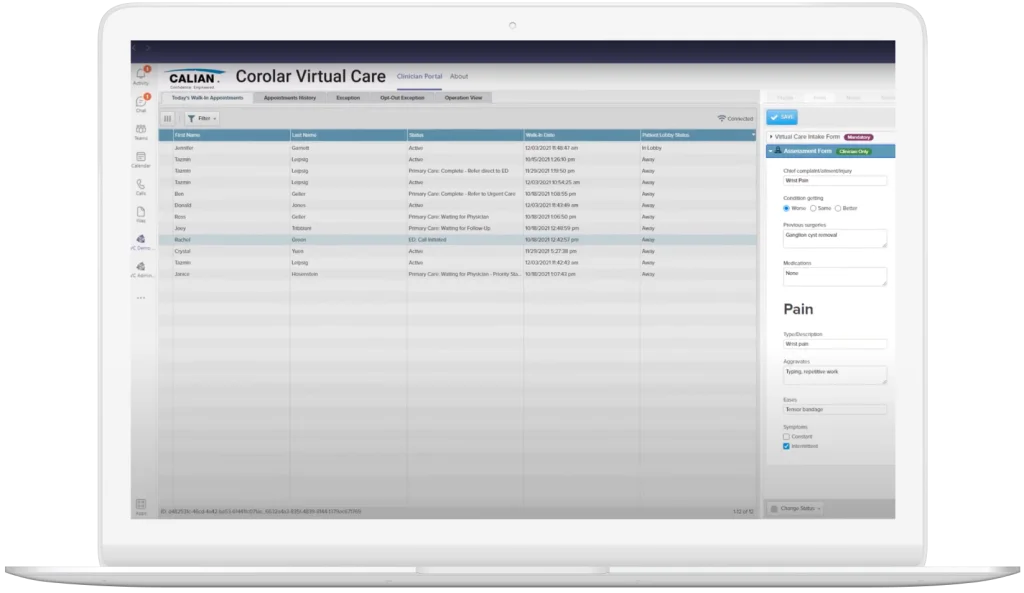
Integrate, connect and deliver—with Corolar
Our Calian Corolar suite of products has been developed to address your need to efficiently integrate systems in a multi-vendor environment without the high costs associated with complex third-party interface development.
We move healthcare forward
We create, develop and manufacture innovative products, solutions, and technologies to solve the worlds most complex challenges.

Explore our innovative healthcare solutions
For more than 20 years, we’ve been providing sophisticated, cost-effective solutions that help people lead healthy lives.